
From clinical trials and studies to
post-marketing surveillance and database studies
―A leading CRO in the evidence generation
with more than 30 years of experience




Building on our experiences in medical care, ArkMS navigates through challenges to open up new horizons in pharmaceutical development.
From pharmaceutical development support to post-marketing, we work closely with people taking on challenges and act as a bridge to deliver reliable data and evidence to future patients and next-generation medical care.
In the ever-changing world of pharmaceutical development and post-marketing surveillance, we disseminate the latest knowledge and findings obtained through projects.
Coming soon.
 Service
Service
We support the entire sequence of your operations, including clinical trials, clinical studies, epidemiological research, post-marketing surveillance (PMS), and safety information management.
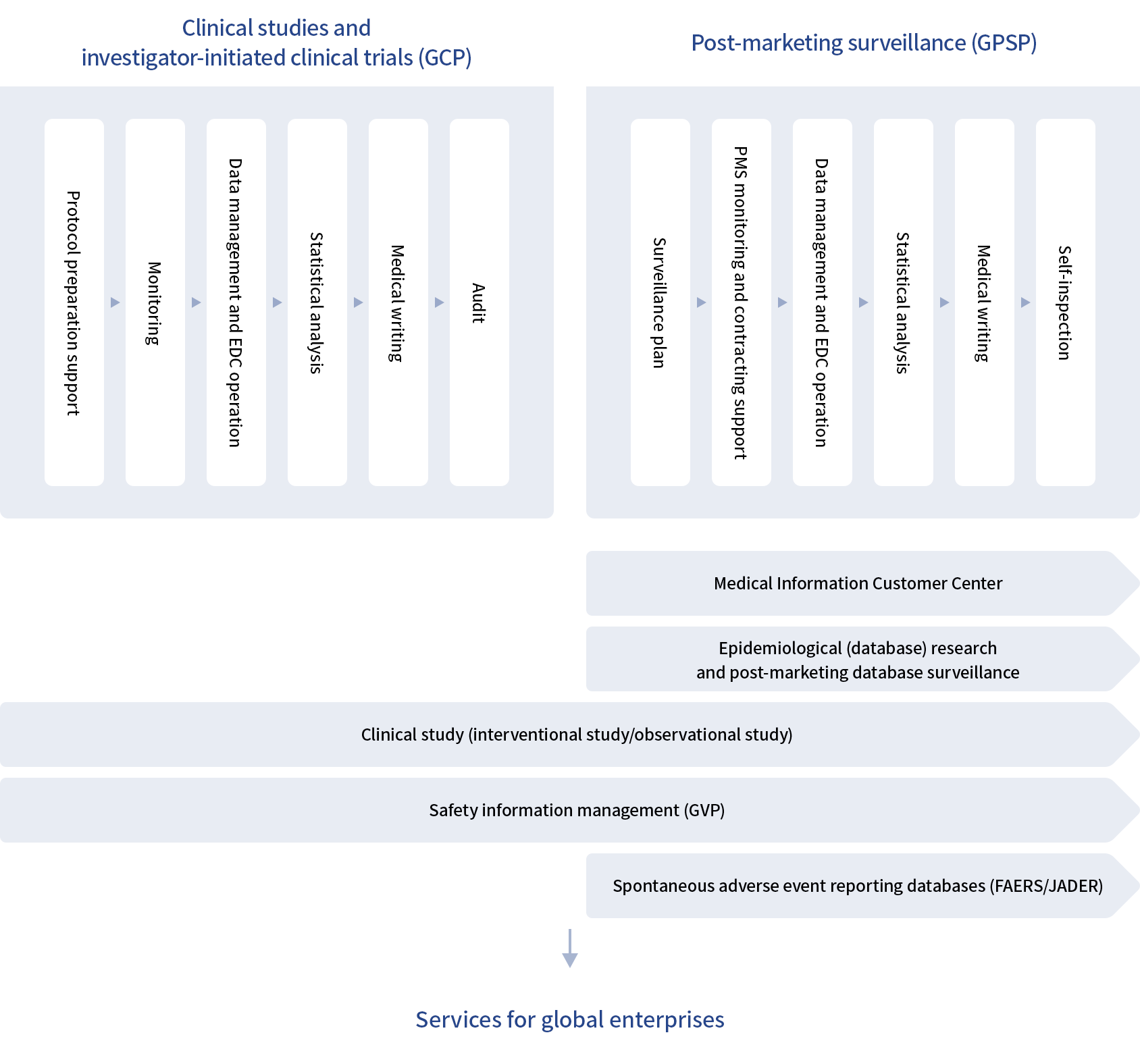

Clinical development support services
We contribute to your success by efficiently conducting clinical trials, including assisting with dealing with authorities prior to the start of clinical trials. We also flexibly handle the establishment of a study system, administrative office support and preparation of protocols, monitoring, DM/EDC, analyses, and clinical study reports for specified clinical trials, observational studies, investigator-initiated clinical trials, academic support, and other operations.
- ・ Support for clinical development (clinical trials/investigator-initiated clinical trials)
- ・ Clinical study support (interventional study, observational study, questionnaire survey)
Post-marketing surveillance (PMS) support services
In addition to the services conventionally provided by CROs in the field of post-marketing surveillance, such as case registration, data management, and statistical analysis, we provide support services for surveillance planning and re-examination application, and many other management operations. We have a system to quickly and accurately respond to various issues that may emerge in post-marketing surveillance.
- ・ Post-marketing surveillance (PMS)
- ・ Safety information management (GVP) support service
- ・ Database surveillance
Other services
We also offer specialized services in various areas such as database surveillance and research, spontaneous adverse event reporting databases, and the Medical Information Customer Center service.
- ・ Medical Information Customer Center
- ・ Database research
- ・ Spontaneous adverse event reporting databases (FAERS/JADER)
Global services
In collaboration with the Alfresa Group companies, we can lead projects as a professional partner providing end‑to‑end support from strategy planning to execution for emerging overseas biopharmaceutical companies seeking to enter the Japanese market.







