Our Global Service
The Most Direct Pathway to the Japanese Market
Alfresa Group’s strength drives your entry into the Japanese market
We partner with innovative biotech ventures and startups, enabling their entry into the Japanese market with comprehensive, end‑to‑end support from strategy planning to execution and solving issues together. Combining the Alfresa Group’s powerful total supply chain service based on a distribution network featuring top wholesale turnover of ethical drugs in Japan with our regulatory and development expertise, we clear Japan’s regulatory and business hurdles to deliver your innovative pharmaceuticals and medical devices to patients without delay.
Why Japan? A promising, world’s third-largest market

Japan stands as the world’s third‑largest healthcare market*, supported by universal health coverage and a high‑cost medical expense benefit system that cater to a wide patient outreach for offering innovative therapies. New fields such as regenerative medicine and digital therapeutics are gaining momentum, supported in a part by government regulatory reforms. Yet for many overseas companies, strict regulations, unique drug pricing rules, and cultural and language differences - drivers of Japan’s drug lag and drug loss - still pose major barriers to entry.
* The International Trade Administration, U.S. Department of Commerce:
https://www.trade.gov/country-commercial-guides/japan-pharmaceuticals
Why ArkMS? Your dedicated team in the Japanese market

Ark Medical Solutions (ArkMS) turns an opaque landscape into a clear roadmap, converting the uncertainty of entering Japan into a confident, executable strategy. We are more than an outsourced service provider. We work with you as your team member to address challenges to ensure a successful Japanese market entry. From market feasibility assessments and Japan‑specific clinical trial design to regulatory interactions, post‑marketing surveillance, safety information management, and the analysis and utilization of real‑world data (RWD), we provide seamless, end‑to‑end support together with our alliance partners. Our ecosystem minimizes Japan‑specific risks and maximizes the value of your business.
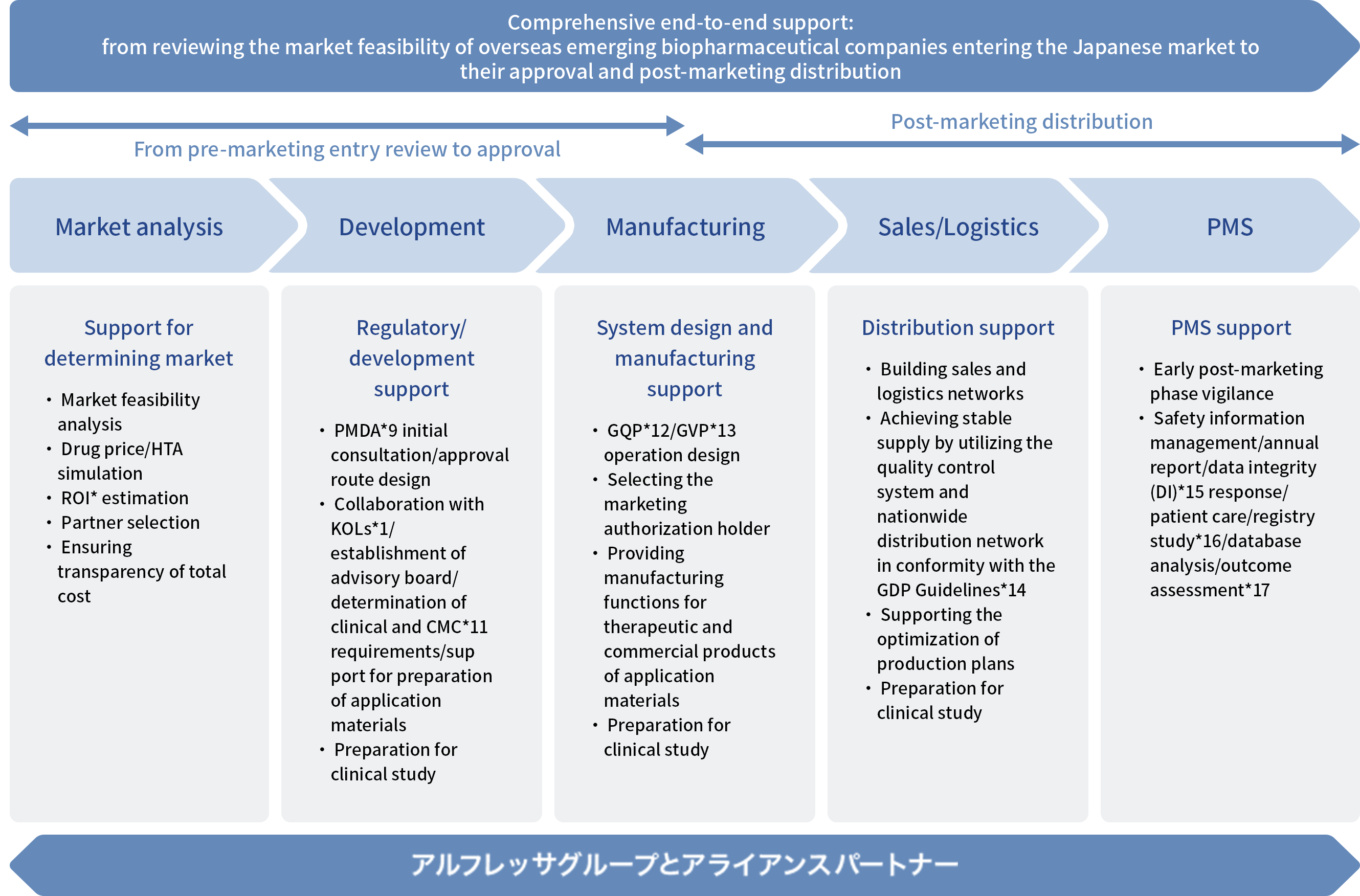
PATH‑Solution: A Platform supporting entry into Japan

The Alfresa Group is expanding and evolving the total supply chain service (TSCS) to meet the needs of Japanese and overseas pharmaceutical companies in a unified manner as a group. By enhancing its functions from introduction, development, and manufacturing of pharmaceutical products, etc., to their distribution, sales, and post-marketing surveillance (PMS), the Alfresa Group serves customers up to the last mile. Utilizing TSCS functions, we provide the services with PATH-Solution, a platform that comprehensively supports your swift entry into the Japanese market, removing barriers for emerging overseas biopharmaceutical companies.
PATH‑Solution is a highly flexible platform that allows overseas emerging biotech and biopharmaceutical companies to combine service modules freely according to their specific needs. Together with Ark Medical Solutions (ArkMS), which has more than 30 years of CRO experience; Alfresa Pharma, which has expanded its contract manufacturing business since 2009; Cell Resources, which is building a globally compliant and reproducible manufacturing framework in regenerative medicine; and the Alfresa Group’s pharmaceutical wholesale business, which operates Japan’s largest distribution network, we establish and continually strengthen an optimal support structure, further enhanced through collaboration with our alliance partners.
January 30, 2026, press release “Launch of PATH‑Solution, a Japan Market Entry Support Platform for Overseas Emerging Biopharmaceutical Companies to Address Drug Lag and Drug Loss”
Source: https://ssl4.eir-parts.net/doc/2784/ir_material17/272468/00.pdf
“PATH-Solution”: https://www.alfresa.com/tscs/path-solution/
ArkMS Service Coverage

 Market feasibility assessment
Market feasibility assessment
Understanding the optimal positioning for entering the Japanese market is a critical first step. Leveraging ArkMS’s experiences and the Alfresa Group’s distribution network and medical services, we evaluate the sales potential and prospects for reimbursement price listing of new pharmaceuticals, medical devices, and regenerative medical products in Japan.
Research themes: Potential/apparent number of patients, current treatment status, reimbursement price listing strategy, optimal clinical trial design, etc.
Research Methods: Desk research (review of public sources), analysis of claims data, interviews with physicians and patients, interviews with other stakeholders, questionnaire surveys, etc.
As part of our market feasibility analysis*, we can also provide rapid reporting on treatment patterns and estimated patient populations by utilizing Japanese claims data owned by AHI partners Inc.*, a member of the Alfresa Group.
*Market feasibility analysis: An analysis of the market size, estimated number of patients, actual treatment status, competition, etc. in the target disease/product area
*AHI partners Inc. https://www.ahi-p.jp/
 Clinical development support
Clinical development support
We also provide support for global clinical trials, including those conducted in Japan. Our team has extensive experience in PMDA consultations, site selection, feasibility assessments, trial preparations, monitoring, and audit operations. In addition, our safety and pharmacovigilance services cover activities such as preparing and managing safety reports during the study.
All services are delivered in compliance with Japanese GCP regulations by a small, highly skilled team that includes locally specialized external experts. This structure enables rapid execution while ensuring seamless communication through team members who are native English speakers.
 Regulatory strategy/approval applications/reimbursement support
Regulatory strategy/approval applications/reimbursement support
In collaboration with alliance partners of the Alfresa Group, we support the development of optimal strategies for pharmaceutical affairs and reimbursement in Japan and support discussions with Japanese regulatory authorities (PMDA* and MHLW*).
*PMDA: Pharmaceuticals and Medical Devices Agency
*MHLW: Ministry of Health, Labour and Welfare
 Post-marketing evidence
Post-marketing evidence
From post‑marketing surveillance to safety information management (GVP), our team provides comprehensive support across a wide range of activities. We also have experience conducting post‑marketing database studies and research, enabling efficient approaches to meet post‑marketing evidence requirements. In addition, we support the design and implementation of long‑term post‑marketing evidence studies - such as additional studies and registry‑based research - to build evidence for future indication expansion applications.
 Other services
Other services
We also provide support for ancillary services required within Japan.
(Ancillary Services)
DB Research
Japan has a well‑established ecosystem for database research utilizing large‑scale claims data and electronic health records (EHR). Our team has extensive experience conducting analyses using the major databases available in Japan and preparing materials and manuscripts for academic conferences and publications.
HEOR/RWE service
While clinical trials are often essential for obtaining regulatory approval, health economics and outcomes research (HEOR) and real‑world evidence (RWE) play a critical role in further establishing product value in the Japanese market. Our team has extensive experience in designing and conducting HEOR and RWE studies tailored to the Japanese market.
Full-scope medical writing
High‑quality research requires the proper development of protocols, statistical analysis plans, clinical study reports (CSRs), and publications. Our team and partner network bring deep expertise in medical writing for studies conducted in Japan.
Partnering/D-MAH support
Establishing a local office in Japan is not always a practical or feasible option when launching a new product in the Japanese market. By leveraging our partner network, we support the selection of optimal manufacturing partners for Japan and provide services through designated marketing authorization holders (D‑MAHs) who can act on behalf of overseas companies in Japan.
ArkMS Key Strengths
- More than 30 years of experience as a CRO in Japan
- Extensive knowledge and abundant experience of RWD in Japan
- Research experience in medical devices, digital therapies, and regenerative medicine/cell therapies, in addition to pharmaceuticals
- We utilize Alfresa Group’s* distribution network as well as medical and related support services in the Group to provide optimal assistance for the specific needs of each client.
* Alfresa Group is a corporate group that operates Japan’s largest distribution network for pharmaceutical wholesale in Japan.





